On Monday, the US National Institute of Health(NIH) announced the initiation of mid-stage clinical trials aimed at testing several treatments for patients experiencing symptoms of long covid. Among the treatments being studied is Pfizer’s Paxlovid, along with at least three other treatments.
The objective of the trials is to assess the safety and efficacy of the treatments in managing the symptoms of long COVID, which refers to the persisting symptoms experienced by patients even after recuperating from the initial phase of COVID-19.
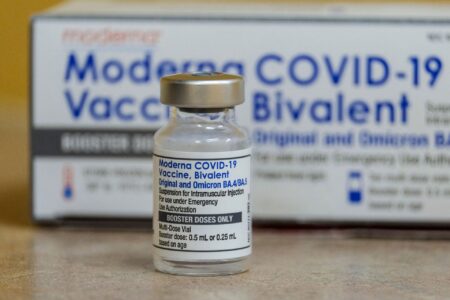
The first part of the trial will specifically focus on testing a longer dosing regimen of Pfizer’s antiviral COVID-19 drug, Paxlovid. The aim is to determine whether this longer dosing regimen can improve the symptoms of long covid in patients.
This study is part of a larger effort to identify effective treatments for long covid, which is a condition that affects a significant number of individuals who have recovered from COVID-19 but continue to experience debilitating symptoms.
Long covid is a complex medical condition characterized by more than 200 symptoms, which can persist for months or even years following a COVID-19 infection. These symptoms range from exhaustion, cognitive impairment, pain, fever, and heart palpitations, among others.
Long covid can be a debilitating condition that significantly impacts the quality of life of those affected. It is a challenging condition to manage, and effective treatments are needed to help individuals recover and return to their normal lives.

NIH Covid
The National Institutes of Health (NIH) is the foremost federal agency responsible for conducting and supporting fundamental, clinical, and translational medical research aimed at uncovering the causes, remedies, and cures for a wide range of common and rare illnesses. The NIH comprises 27 Institutes and Centers and operates as a division of the United States Department of Health and Human Services.
The National Institutes of Health stated that in addition to the aforementioned aspects, other components of the study will assess symptoms associated with cognitive dysfunction such as brain fog and memory impairment.
Medical interventions such as a web-based brain training program named Brain HQ, developed by the San Francisco-based private company Posit Science Corporation, and a device by Soterix Medical Inc. that utilizes a small electric current to stimulate brain activity, will be included in the study.
The National Institutes of Health stated that they anticipate undertaking further clinical trials to evaluate at least seven additional treatments.
The trials being carried out as part of the Researching COVID to Enhance Recovery (RECOVER) Initiative by the NIH are informed by previous RECOVER research conducted over the last two years, and concentrate on addressing several of the symptoms that are identified as particularly distressing by individuals suffering from long COVID.
The RECOVER initiative’s coordinated research endeavors have enabled the NIH to design and execute trials that have the potential to alleviate the symptoms of long COVID, which can vary widely, more expeditiously than any individual study could achieve on its own.
Statements by NIH
Acting NIH Director Lawrence A. Tabak, D.D.S Ph.D stated that they understand the urgency of addressing the suffering experienced by patients, and that the NIH is dedicated to employing a meticulously planned and scientifically sound strategy to identify treatments that can alleviate the symptoms of long COVID for the millions of individuals affected.
Walter J. Koroshetz, M.D., director of the NIH’s National Institute of Neurological Disorders and Stroke and co-lead of the RECOVER Initiative, stated that hundreds of RECOVER researchers and participants are diligently investigating the biological underpinnings of long COVID, a condition that can impact nearly all bodily systems and is characterized by over 200 symptoms.
Acknowledging the likelihood that multiple solutions may be required, the RECOVER Initiative has utilized insights gleaned from study participants to design robust clinical trial frameworks that can identify treatments for individuals with varying symptom profiles, in order to enhance their overall well-being and functionality.
The trials that commenced today will specifically target viral persistence and cognitive impairment, utilizing “platform protocols,” a phrase that refers to the flexible design of these trials.